Answers and suggestions for the topic-based activities
Activity 1 - The scientific method:
- Try to suggest key terms such as observation, hypothesis, prediction, testing, confirm, falsify.
- Hopefully some parts of your flowchart match with those in the scientific method.
- Repeatability: Hopefully, to confirm your hypothesis you watched a number of turns (repeated outcomes) to add strength to your suggestion. Ideally you might ask students in a different class to also confirm it. Falsification: The strength of our hypothesis can be better tested by actively looking to disprove it.
- In science, there is no one to tell us whether or not our hypotheses are 100% correct or not so we can never be completely certain.
Activity 2 - The role of reason in the natural sciences:
- C.
- See reasoning here.
- This is inductive reasoning - the formation of a general trend or pattern from a range of individual points.
- The natural sciences produce knowledge by identifying patterns from empirical evidence. We can test the reliability of these patterns by making predictions and then testing them experimentally.
- Unless we have 100% of the data, we can never be 100% sure that a pattern exists. Practically speaking, collected an infinite number of data points is impossible.
Activity 3 - Ways of knowing in the natural sciences
- Rutherford's gold foil experiment (17.50-25.00): Sense perception - Required to collect data of the marks seen on the zinc sulfide detection screen; intuition - asking the research assistant to move the detection screen to the same side as the radioactive source suggests Rutherford's may have had a feeling that they were not seeing the full picture; Reason - Rutherford used logic to suggest an explanation for his experimental results e.g. as only a very small percentage of the alpha particles were reflected, the thing they are hitting (the nucleus) must take up very little space in the material.
- Mendeleev's predictions of the missing elements: Reason - Finding evidence of atoms with a large range of number of protons indicated that there would likely be atoms with all numbers of protons (up to around 100 at that time). This would be an example of inductive reasoning; Faith - Some scientific knowledge in Mendeleev's day relied on the belief that these gaps would one day be filled.
- Kekulé's dream about the structure of benzene: Intuition - If the claim about the dream was true, it may suggest that Kekulé's prior work with chemical structure may have given him unconscious insight; Imagination - dreaming might indicate the role of imagination and divergent thinking in solving scientific problems.
- Fritz Haber's work on chemical weapons: Emotion - Haber was driven to this research via his patriotic sentiment although this gas would be later used on his fellow Jewish citizens.
- Organic nomenclature: Language - Given the immense range of organic compounds, an internationally agreed use of language to describe them is essential.
Activity 4 - Mystery box
- -
- Likely a lot of sense perception (holding/listening), reason and imagination. Language, intuition and memory also may play a significant part. Emotion and faith may be less important.
- Not necessarily. Depends on the use of the box. The model may be better for predicting the behaviour of the box.
- Weighing, use of magnet, float testing, x-ray.
Activity 5 - Natural science v religion
- What kind of knowledge does it produce? NS: Descriptions and proposed explanations for how the natural world works. R: Meaning around why we are here and frameworks for how we should live our lives; some religions may suggest explanations for how we got here.
- What kind of evidence is used? NS: Empirical evidence - Data collected from experiments (sense perception important here). R: Interpretation of religious texts and experiences. Perhaps some physical evidence such as the Dead Sea scrolls.
- What might certainty mean? NS: May relate to closeness of theory to empirical evidence; may connote the accuracy of predictions; may relate to success in peer review process. R: May relate to an individual's intuitive senses; may relate to agreement between religious scholars: may relate to literal truth of religious texts.
- What is considered reliable knowledge? NS: Knowledge that is replicable and cannot be falsified; predictive accuracy. R: Knowledge that is agreed upon by many.
- Who would be considered an expert in it? NS: A scientist; editor of scientific media R: A religious scholar or leader.
Topic 1 - Stoichiometry
Activity 1 - Phlogiston: The mass of the magnesium increases during combustion as magnesium oxide is formed. This contrasts the idea that burning something releases the phlogiston into the air.
Extension: Joseph Priestley and Antoine Lavoisier worked in the 1770´s on the idea that some materials gained mass when reacted with air.
Activity 2 - Unit convention:
- 1000Mm --> 1000 Mm
- 5 Candela --> 5 candela
- 25.06 ccd --> correct
- 94 K --> correct
- 74888 m seconds --> 74888 ms OR milliseconds
- 0.004Moles --> 0.004 moles
- 20 hm --> correct
- 12 μ m --> 12 μm
- 499 Mega seconds --> 499 megaseconds
- 2 nano Kelvin --> 2 nanokelvin
Extension: Alongside more complex reasons, the kg was a more practical unit to use in terms of trading goods.
Activity 3 - Theory or law?: Following phrases are most suitable:
- Is a mathematical statement about phenomena.
- Is rarely adapted.
- Is assumed to be scientific truth within its parameters. (i.e for an 'ideal' gas)
- Is not developed from a theory. (as it does not offer an explanation)
- Can be used to make predictions.
Activity 4 (TOK) - The significance of laws in chemistry:
- See link.
- This would alter one of the most fundamental truths in chemistry (and physics). We would have to explore where mass goes or is created from. It might open many exciting avenues for research. It might mean that all industrial chemical process are reviewed as stoichiometry as we know it would become redundant.
- The scientific method requires replication of results. If replication was not possible then the data would not be considered reliable.
Activity 5 - Gas laws and innovation: As well as supplying some air flow into houses, the air is initially compressed as it enters the narrow part of the bottle but when it undergoes sudden expansion as it leaves the narrow opening and enters the house, it cools down. See video of the project here - although the explanation seems wrong as it suggests the air cools as it is compressed.
Topic 2 and 12 - Atomic structure
Activity 1 - How did Germany let its nuclear capabilities slip away before WWII: These scientists were all Jewish and contributed to the field of nuclear science in and around Germany during the early/mid 1900´s (the time of the Second World War). They all left Germany due to the persecution of the Jews during this period. They were some of the leading minds in this field and, had they of stayed in Germany, perhaps the Germans would have been able to develop nuclear weapons before the Allies.
Extension: The "Manhattan Project" was initiated in 1942 to develop nuclear weapons before the Germans. It was a collaboration between the US, UK and Canada.
Activity 2 - The 4 new chemical elements: N/A
Activity 3 - The 5th Solvay Conference: Werner Heisenberg (came up with the uncertainty principle), Louis de Broglie (described wave-particle duality) and Ernest Schrödinger (wrote the Schrödinger equation).
Women were heavily excluded from further education until the 1900s so there were few prominent figures in science at that time.
Women were heavily excluded from further education until the 1900s so there were few prominent figures in science at that time.
Activity 4 - TOK - Atomic modelling: How do we know?: Sense perception - viewing the flashes on the photosensitive shield; Intuition - the presenter suggests Rutherford had 'a feeling' that something else was happening; Reason - using logic to explain why only 1 in 8000 positive alpha particles passed through the gold sheet.
Topic 3 and 13 - Periodicity
Activity 1 - The 4 new elements: Suggestions.
All elements - Largest atomic radius in respective groups.
Element 113 - Metal. Common oxidation state of +3. Insoluble oxide.
Element 115 - Metal. Common oxidation states +5, +3, -3. Acidic oxide.
Element 117 - Non-metal. Solid. Would it be found as a diatomic molecule? If so then higher boiling point than others in group 17.
Element 118 - Non-metal. Colourless, odourless, monoatomic gas.
All elements - Largest atomic radius in respective groups.
Element 113 - Metal. Common oxidation state of +3. Insoluble oxide.
Element 115 - Metal. Common oxidation states +5, +3, -3. Acidic oxide.
Element 117 - Non-metal. Solid. Would it be found as a diatomic molecule? If so then higher boiling point than others in group 17.
Element 118 - Non-metal. Colourless, odourless, monoatomic gas.
Activity 2 - Ytterby: Yttrium, ytterbium, terbium, and erbium.
Extension: Holmium and Gadolinium (after Johan Gadolin)
Activity 3 - Chemical weapons: Opinion based so no correct answers.
Extension: Chlorine undergoes hydrolysis with water. Cl2 + H2O --> HCl + HClO
Topic 4 and 14 - Chemical bonding and structure
Activity 1 - Metallic hydrogen???: Extremely high pressures to force H atoms into a solid lattice with possibility of delocalised electrons.
Activity 2 - The most simple Nobel Prize yet?: The story of the discovery of graphene can be found here.
Activity 3 - The 1987 Montreal Protocol: Possible consequences --> Higher skin cancer rates, higher levels of blindness, large scale damage to vegetation.
Activity 4 - Calming waves on a lake using olive oil: LINK to full explanation video. Olive oil is non-polar and contains only London forces whereas water is very polar and contains all three intermolecular forces.
Activity 5 - What really causes bonding?: Example: Covalent bonding --> Why? --> Two nuclei are attracted to a shared pair of electrons --> Why? --> Nuclei are positively charged and electrons are negatively charged. Opposite charges attract --> Why? --> Coulomb's Law --> Why? --> Mathematical modelling in this case is effective in explaining observable phenomena --> Why? --> No idea! We end up maths (an abstract system that humans invented) which is used to explain electrostatic attraction and potential energy. These seem to be the starting points to our ability to explain bonding.
Topic 5 and 15 - Energetics
Activity 1 - The limitations of models:
a. Lithium iodide has a greater lattice enthalpy as the lithium ion has a smaller atomic radius and therefore a greater charge density and stronger attraction to the iodide ion.
b. Silver iodide has a large lattice enthalpy.
c. We would expect the smaller sodium ion to lead to a greater lattice enthalpy.
d. The difference must be caused by an additional degree of covalency found in the silver iodide that is not considered using the ionic model.
a. Lithium iodide has a greater lattice enthalpy as the lithium ion has a smaller atomic radius and therefore a greater charge density and stronger attraction to the iodide ion.
b. Silver iodide has a large lattice enthalpy.
c. We would expect the smaller sodium ion to lead to a greater lattice enthalpy.
d. The difference must be caused by an additional degree of covalency found in the silver iodide that is not considered using the ionic model.
Activity 2 - Hess's law and accuracy
1. Hess's law is an application of the law of the conservation of energy. True
2. Experimentally, DeltaH1 will be exactly the same as DeltaH2+DeltaH3. False (very unlikely)
3. Experimental uncertainty may affect the accuracy of using Hess's law. True
4. The number of reactions used in an indirect pathway would decrease the total % uncertainty in the process. True (the more measurements made, the more uncertainty to take into account)
5. The data found in the data booklet has no uncertainty. False (there is none written, but all experimental measurements have uncertainty)
6. Data in the data booklet is exactly the same as other literature sources. False (linked to previous answer)
7. A scientific law can be disproven. True (if new evidence does not fit the law then we may have to adjust or make exceptions)
1. Hess's law is an application of the law of the conservation of energy. True
2. Experimentally, DeltaH1 will be exactly the same as DeltaH2+DeltaH3. False (very unlikely)
3. Experimental uncertainty may affect the accuracy of using Hess's law. True
4. The number of reactions used in an indirect pathway would decrease the total % uncertainty in the process. True (the more measurements made, the more uncertainty to take into account)
5. The data found in the data booklet has no uncertainty. False (there is none written, but all experimental measurements have uncertainty)
6. Data in the data booklet is exactly the same as other literature sources. False (linked to previous answer)
7. A scientific law can be disproven. True (if new evidence does not fit the law then we may have to adjust or make exceptions)
Topic 6 and 16 - Kinetics
Activity 1 - The Contact Process: Suggestions - Higher production in wealthy countries/industrial nations/northern hemisphere/large cities
Extension: Vanadium(v) oxide. Transition metals can have a range of oxidation states so are effective catalysts.
Topic 7 and 17 - Equilibrium
Activity 1 - Shade balls:
- Dissolution of chemicals from balls into water (especially given high temperatures) - effect on human health and environment.
- Cost of balls v cost of evaporated water.
Extension: Original purpose was to prevent oxidation of chloride (Cl-) and bromide (Br-) ions to harmful ions such as the carcinogenic bromate ion (BrO3-). Relates to Topic 9 and 19 - Redox processes.
Activity 2: The application of science: Source 1a refers to his Nobel prize for his advancement of the process of creating ammonia, a much needed chemical for fertilisers. Source 2 refers to his work developing chemical weapons for Nazi Germany.
Extension: Nuclear physics - gave rise to the possibility of nuclear energy as a resource but also the atomic bomb. Agent Orange - A successful fertiliser for soybeans but used as a chemical weapon in the Vietnam War on an estimated 400,000 people. Trinitrotoluene (TNT) - Originally developed as a yellow dye until its explosive properties were exploited. Freon (a CFC) - A safer alternative to other refrigerants at the time but became a major cause of ozone depletion. Lead in petrol - Substantially increased engine performance in cars but likely responsible for huge numbers of deaths due to lead poisoning.
Topic 8 and 18 - Acids and bases
Activity 1 - Acid rain: N/A
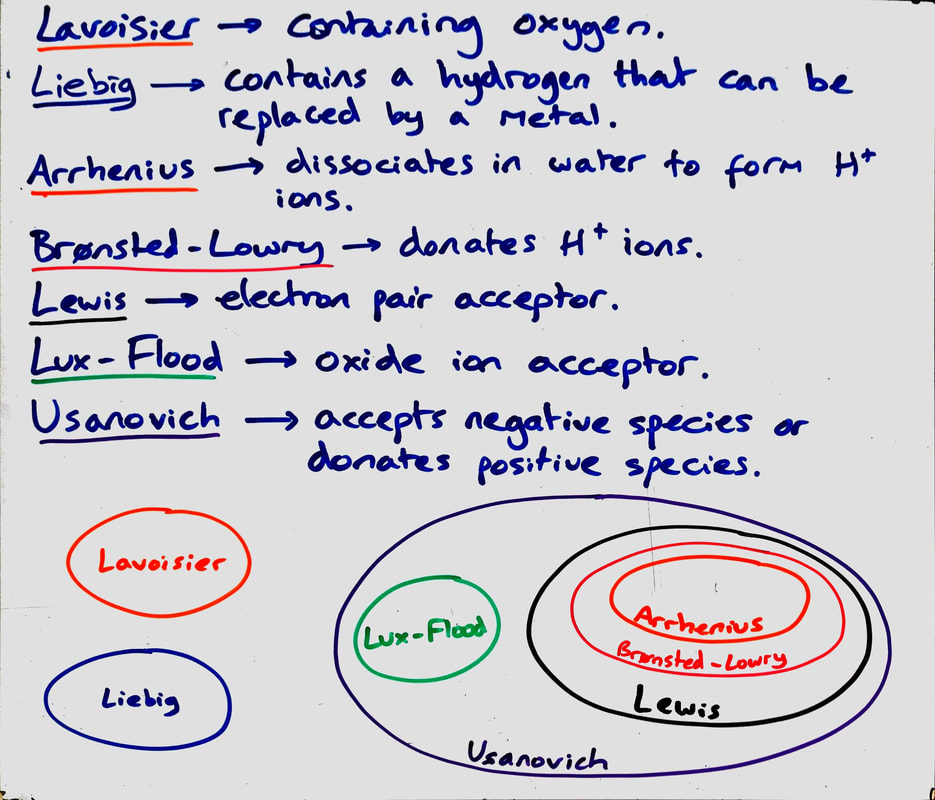
Activity 2 - Theories of acids:
Possible answers:
A - Acids must contain H and O/only nonmetals/only H and Group 15 and 16 elements.
B - Acids must contain H and Group 14-17 elements. Oxygen is not required to be an acid.
C - Acids can be charged species.
D - Acids do not need to have H. Acids can have Group 13 elements.
Possible answers:
A - Acids must contain H and O/only nonmetals/only H and Group 15 and 16 elements.
B - Acids must contain H and Group 14-17 elements. Oxygen is not required to be an acid.
C - Acids can be charged species.
D - Acids do not need to have H. Acids can have Group 13 elements.
Extension: See links or wikipedia page. Essentially acids that cannot be included on the pH scale as they are too strong.
Topic 9 and 19 - Redox processes
Activity 1 - Animal electricity:
- The nervous system in animals.
- He increased the voltage provided by this process by building up multiple, alternating layers of the two metals.
- Animal electricity refers to the process of muscle contraction due to electrical currents in an organism.
Activity 2 - Who cares?:
- Use of fuel cells could reduce harmful emissions - better for human health, fewer negative environmental consequences.
- Renewable sources of energy are now a large political issue e.g. US elections.
- Specific photo relate to global warming.
- Cost of research and implementation of new fuel technologies can impact national budgets, job production etc.
- Are we morally obliged to protect our planet against pollution and climate change? Interesting LINK.
Topic 10 and 20 - Organic chemistry
Activity 1 - Cause vs correlation:
- 1st graph: 1. Yes, fairly clear correlation; 2. E.g. More imported oil requires more transportation --> more trains used for transportation --> higher likelihood of collisions; 3. Some explanations could probably be considered to provide a mechanism but the number of unknown variables would appear to make it unlikely.
- 2nd graph: 1. It would be more difficult to explain this apparent correlation with a sensible mechanism; 2. Unlikely; 3. Hopefully this exercise that a correlation is not sufficient to prove a causal relationship.
Activity 2 (TOK) - Ways of knowing in chemistry: The story suggests a dream was responsible so we might categorise that as imagination and intuition. Sense perception and reason are perhaps the most widely used ways of knowing in chemistry.
Topic 11 and 21 - Measurement and data processing
Activity 1 - Faster than the speed of light: 1. 15000 repeats were made; 2. The error must be systematic as the error was the same size and carried in the same direction each time; 3. Scientists were sceptical about the results. The reliability of the results were tested by repeat measurements at another facility. These measurements showed results that were consistent with Einstein´s special theory of relativity.
Activity 2 - Bike safety: Relevant links: LINK 1, LINK 2.
1. Suggestions: Cyclist take more risks as they feel safer; Cars around the cyclist take more risks as the cyclists appear safer; Less able/confident cyclists more likely to wear helmets but still more prone to accidents.
2. Use a sample group of a 50:50 split of helmet wearing/non-wearing cyclists who cycle in a similar area of a city over a certain period of time. Record number of accidents experienced and compare them.
3. Ability of cyclists; amount of cycling done by individuals; exact routes takes; reliability of bicycles etc.
1. Suggestions: Cyclist take more risks as they feel safer; Cars around the cyclist take more risks as the cyclists appear safer; Less able/confident cyclists more likely to wear helmets but still more prone to accidents.
2. Use a sample group of a 50:50 split of helmet wearing/non-wearing cyclists who cycle in a similar area of a city over a certain period of time. Record number of accidents experienced and compare them.
3. Ability of cyclists; amount of cycling done by individuals; exact routes takes; reliability of bicycles etc.
Option A - Materials
Activity 1 - The presentation of scientific findings and issues: Possible considerations - Electronics, nanorobots, medicine, communications, unknown consequences on health and environment...
Activity 2 - The discovery of polywater: 1 - There may be some introduction of random error in his experiment; an uncontrolled variable may be influencing his results. 2 - He could have checked the purity of the initial water; check the function and cleanliness of the equipment.
Option B - Biochemistry
Activity 1 - What to eat?: No specific answers.
Extension: Government - ; Food industry - profit losses/gains if specific products are discouraged or encouraged; You - Economic considerations, lifestyle choices, friends and family; Medical profession - Area of expertise may affect knowledge, doctors can be paid by certain industries to promote specific lines of thinking; Media - Higher profit from sensationalist stories, levels of scientific knowledge.
Activity 2 - Is 'chemical surgery' acceptable?:
- Benefits: Genetic disorders could be eliminated, eliminating human suffering in those areas.
- Opponents: Those with some existing genetic disorders may argue that the idea devalues their experience. Religious groups may argue against interference in 'natural' order. High initial prices will limit advantages to the wealthy.
- Difficult question!
Option C - Energy
Activity 1 - Sexism in science - Lisa Meitner: Opportunities in higher education for women were extremely sparse. Women were refused access to basic research facilities for many years as seen in the case of Lise Meitner. Even the Nobel committee was heavily prejudiced against the achievement of women.
Extension: Meitnerium (Mt) 109.
Activity 2 - Public concern with nuclear energy: Lack of understanding leads to exaggerated views of risks in terms of nuclear explosions, radiation problems etc. Needs to be weighed against the current impact of burning fossil fuels. Not helped by publicity around infrequent 'disasters' such as Chenobyl and Fukishima. Many countries already rely quite heavily on nuclear - the US gets around 20% of its energy from it.
Activity 3 - Accidentally the most environmentally destructive man in history: Scientific discoveries are morally neutral in themselves but the applications can lead to unintended consequences.
Activity 4 - Nike and global warming
- Why did Nike stop using the sulfur hexafluoride? It was found out to be a very potent greenhouse gas (25,000 times worse than carbon dioxide) and remained in the atmosphere a long time due to it being very unreactive.
- What evidence is there for the problems caused? Data from ice cores, tree rings, sediment samples; temperature data since recordings began; atmospheric composition testing; sea level measurements; ice cap measurements.
- Can you suggest any ideas why some people might not 'believe' in global warming? Political bias; reference to solar cycles or other natural fluctuations in global temperatures; potential for bias in research grants;
Option D - Medicinal chemistry
Activity 1 - A lucky discovery?: Possible examples: Viagra; Microwave background radiation; Safety glass; Saccharine; LSD; Bakelite; X-rays; Teflon; Gunpowder
Activity 2 - The dangers of clinical drug testing:
- Biological testing in discovery research phase; Increasingly sized human groups in developmental trial phase; Post-marketing monitoring
- The fault is debatable. The system is designed to minimise risk but 100 % safety is unrealistic as the effects of new compounds can be unpredictable. There is sometimes criticism that a majority of those who put themselves forward for the clinical trials are more likely to financially vulnerable (e.g. students and the impoverished) and less aware of the possible risks.
- Animal testing is a highly emotive subject.
- Advantages: Reduces negative consequences on humans in clinical testing; Arguably more effective than in vitro testing. Animals themselves benefit from many of the products being tested. Shorter life cycles of animal make them a more effective subject.
- Disadvantages: Ethically acceptable/Inhumane?; Animal bodies do not always respond to drugs in the same way as with humans so some testing may be unnecessary or misleading; Should testing be allowed for products that are not medicinally beneficial i.e. makeup?; Expensive;
- Advantages: Reduces negative consequences on humans in clinical testing; Arguably more effective than in vitro testing. Animals themselves benefit from many of the products being tested. Shorter life cycles of animal make them a more effective subject.
Activity 3 - Public understanding
1. Mild analgesics all act using the same mechanism of preventing interaction between prostaglandins and the pain receptors at the site of pain.
2. Mild analgesics act at pain receptors in all areas of the body so they cannot be said to target the area when pain is being felt.
1. Mild analgesics all act using the same mechanism of preventing interaction between prostaglandins and the pain receptors at the site of pain.
2. Mild analgesics act at pain receptors in all areas of the body so they cannot be said to target the area when pain is being felt.
Extension: Nurofen Express tablets contain the sodium salt of ibruprofen so are more soluble in the blood and therefore act more quickly.